Chemical Education Journal (CEJ), Vol. 14, Issue 1 /Registration No. 14-3 /Received October 6, 2011, Revised November 24, December 21.
URL = http://chem.sci.utsunomiya-u.ac.jp/cejrnlE.html
Corresponding author: Hidetsugu KOHZAKI, PhD, Department of Cell Biology, Institute for Virus Research, Kyoto University, Shogoin-kawahara-machi 53, Sakyo-ku, Kyoto, Japan 606-8507.
E-mail: charaznable.k@gmail.com
Keywords
Chemical education, Medical technologist, Training institutions
Total number of words: 22,900
Short running title: Chemical education for medical technologists
We teach chemistry at a medical technology school. Chemistry is not necessarily the favorite subject of Japanese students receiving paramedical education. However, many biochemical, microbiological, and advanced genetic tests are performed in the medical setting, and solutions and media also have to be prepared. Nurses and paramedics including medical technologists rarely prepare solutions because of automation in the medical setting and a lack of time. However, they might feel uneasy about certain aspects of medical practice, such as the use of infusions and injections, if they do not know how to prepare and label solutions. In addition, the annual national examinations include questions on concentration calculations.
In this article, we introduce our approach to providing chemical education for medical technologists.
In Japan, chemistry acted as the main driver of growth during the high-growth period. Thus, at that time, chemical education supported Japan. However, the academic ability of Japanese students has declined markedly because of the trend towards "science phobia" and pressure-free education; e.g., teaching the value of "pi" as 3 and not requiring students to memorize the periodic table of elements. Since the introduction of IT, information science has been required in various fields [1]. However, this is not true of chemistry. Recently, many Japanese researchers have won the Nobel Prize. Thus, science education, especially chemical education, has come to be reevaluated. We teach chemistry at a medical technology school. Although many of our students are good at IT, they lack chemical knowledge. Japanese junior high school students must choose between humanities or science courses. However, adequate chemical education is provided at junior high school. Thus, even students who enroll in a humanities course should be able to answer the chemical questions on the National Center for University Entrance Examinations. However, the students on the humanities courses stop studying chemistry because their main goal is to pass the college entrance examination. Thus, chemistry is only studied at junior high school by the students who take science courses. In addition, some students lose interest in chemistry because of their desire for pressure-free education.
Thus, chemistry has become unfamiliar to many students entering paramedic schools, such as nurses and medical technologists. In fact, many students have not seen the periodic table of the elements (ref.[2] Chapter 1 I, Table.2 Science I "Basic of chemistry", Chemistry "Chemical bond ") or cannot write the molecular formulae of water and/or carbon dioxide (ref.[2] Chapter 1 I.5, Table.2 Science I "Basic of chemistry ", Chemistry "Chemical bond "), much less the structural formulae of covalent (ref.[2] Chapter 1 II.9, Table.2 Science I "Basic of chemistry ", Chemistry "Chemical bond ") or ionic bonds (ref.[2] Chapter 1 II.8, Table.2 Science I "Basic of chemistry ", Chemistry "Chemical bond ") or amino acids (ref.[2] Chapter 5 IV.2, Table.2 Science I "Amphoteric electrolyte ", Chemistry "Chemistry of proteins").
However, paramedics should have some knowledge about amino acids (ref.2 Chapter 5 IV.2, Table.3 Science I "Amphoteric electrolytes ", Chemistry "Chemistry of proteins "), sugars (ref.[2] Chapter 5 IV.1, Table.2 Chemistry "Chemistry of sugars "), lipids (ref.[2] Chapter 5 IV.3, Table.2 Chemistry "Chemistry of lipids "), ions (ref.[2] Chapter 1 II.8, Table.2 Chemistry "Chemistry bond "), and osmotic pressure (ref.[2] Chapter 2 V). Laboratory test values should be correctly understood by all medical practitioners because they will encounter a wide variety of test parameters during their jobs, such as measures of in vivo enzyme function (enzymatic activity in blood is affected by metabolite levels and diseases) (Table.2 Science I "Enzyme reaction ", Table.4 1.B. "Unit of enzyme "), and the shortage of physicians in Japan is increasing the burden placed on medical institutions. In addition, there is also an increasingly broad range of medicines in use because of the rapid development of novel agents, and numerous diagnostic kits have also been developed. Furthermore, since the completion of the human genome project, chromosomal gene tests have become more common, and pharmacogenomics and personalized medicine have advanced. As a result, chemical questions have arisen on the National Examination for Paramedics (Table.4).
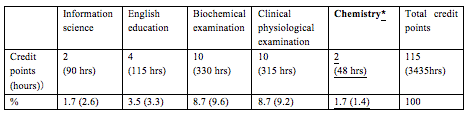
Medical technology schools produce curricula that help their students to pass the National Examination for Medical Technologists. Hence, they provide technical knowledge and skills, but allow no time for chemistry (Table 1). As such, students do not learn the chemistry they will require during medical practice. In addition, students cannot learn all the contents of basic textbooks for medical technology schools [2]. Thus, an exam was conducted to evaluate the basic scientific knowledge of our students. This written exam included questions pertaining to the measurement of acids and alkalis with litmus paper (ref. [2] Chapter 3 IV), the three states of water (ref. [2] Chapter 2, I, II, III), the chemical formula for water (ref. [2] Chapter 1 I. 5), and how to calculate the percentage concentration of NaCl (ref. [2] Chapter 2 IV. 1). It was used to assess whether the students possessed academic knowledge that they were supposed to have learnt during middle school.
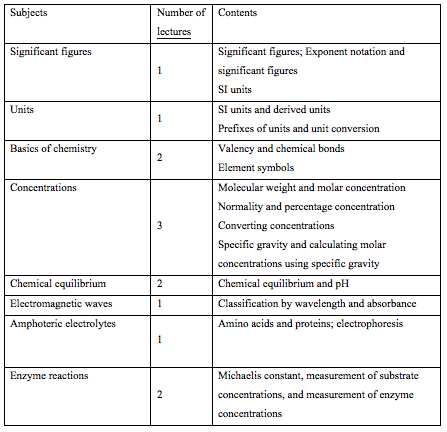
In fact, about 20% of the enrolled students answered less than 50% of the questions in the science and chemistry tests correctly. Most of the enrolled students did not possess chemical knowledge because they did not take chemistry as an elective subject in high school due to a desire for pressure-free education. Thus, we used Mendeleev's periodic table to make them familiar with the chemical world (Table 2). In Science I, we taught chemical bonds, molar concentrations, solution preparation, specific gravity, etc. Colorimetry is important because it is used for the measurement of protein and nucleic acid concentrations, immunostaining, etc., in biochemistry (Table 2A). Thus, chemical knowledge is necessary to understand biochemistry. we also taught enzyme reactions and alcohol metabolism in the living body as well as the in vivo behaviors of proteins and sugars (Table 2B).
A. Science I
Implementation term: First semester of the first year
Time: 18 hours
Teaching method: Lectures
Number of units: One unit
Contents: Exponent notation and significant figures
Molecular weight and concentrations
Syllabus
B. Chemistry
Implementation term: First semester of the first year
Time: 30 hours
Teaching method: Lectures
Number of units: One unit
Contents: Atoms and molecules; bonds; structures of carbon compounds and various linking groups; and the chemistry of sugars, lipids, and proteins
Syllabus

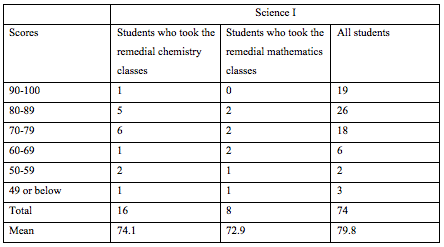
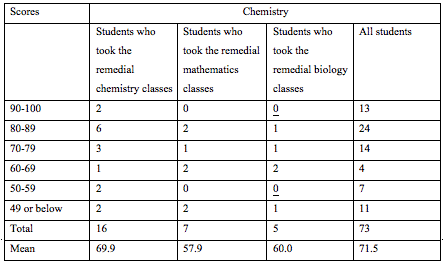
The education program was structured as follows (Remedial classes were introduced in 2008. Students who received these classes took the 55th or later National Examination.)
A
B
The range of possible questions for the National Examination for Medical Technologists (5) is shown in Table 4, and some chemical questions are included on the test. In addition, chemistry is essential for clinical chemistry. Thus, chemistry is a fundamental subject that can have a broad influence on students' education.
Japanese medical institutions have undergone considerable automation, and most test parameters are now automatically examined in clinical laboratories. In addition, the ISO15189 [6] guidelines have been established for clinical laboratories, and the standardization of test data has also been demanded. The following international standards are relevant to clinical laboratories: ISO15193 (In vitro diagnostic medical devices -- Measurement of quantities in samples of biological origin -- Requirements for content and presentation of reference measurement procedures) [7], ISO15194 (In vitro diagnostic medical devices -- Measurement of quantities in samples of biological origin -- Requirements for certified reference materials and the content of supporting documentation) [8], ISO17511 (In vitro diagnostic medical devices -- Measurement of quantities in biological samples -- Metrological traceability of values assigned to calibrators and control materials) [9], and ISO18153(In vitro diagnostic medical devices -- Measurement of quantities in biological samples -- Metrological traceability of values for catalytic concentration of enzymes assigned calibrators and control materials) [10]. ISO9001 (quality management systems) [11] is also applicable. Thus, it is vital that medical technologists are able to produce accurate test data.
Considering the trends in exams over the past 7 years, a mean of 32 chemical questions, including biochemical questions, are set annually as clinical chemical questions in the National Examination for Medical Technologists [5]. However, chemistry includes the fields of biochemistry and clinical chemistry (Table 4). To become a medical technologist, students must pass the national examination, which requires a high score.
We educate medical technologists and have prepared curricula that promote the understanding of biochemistry and clinical chemistry (Table 2). Correctly calculating the effects of our curricula is difficult. However, the results of the National Examination for Medical Technologists show that the mean score for Clinical Chemistry was 10% higher than those for Genetic Testing and Information Science, which we were also responsible for [12] (Table 5), but 4% lower than that for Biology & Biochemistry.
The authors have been offering a remedial chemistry course because it is difficult to encourage students to undergo the basic course for medical technologist training institutions due to the recent reduction in students' interest in science and their lack of scientific knowledge. In fact, some students withdrew from the school because they could not keep up with the class, and the pass rate for the National Examination had begun to decrease (Tables 5 and 6). Although it has been only a few years since the remedial classes were adopted (Table 3), the National Examination scores of our students have increased by 4.4% for clinical science and 2.1% for all subjects, and the pass rate was also 4.2% higher among the students who took the remedial classes than among those who did not. The "Science I " and "Chemistry " curricula that we introduced are not the only the reasons for the increased pass rate; however, it is considered that increases in the students' scores for clinical chemistry contributed to it (Table 6). Basic chemistry provides a solid foundation for further studies in related sciences including clinical chemistry and gene testing, particularly biology and biochemistry.
Therefore, we've admitted that our program was not a complete success; however, it seems to have helped the students to pass the National Examination (Figure 1, Table. 6).

Furthermore, qualifications, such as the Qualified Class 1 and 2 Laboratory Technologist qualifications [14] and the Japan Society of Clinical Chemists qualification [15], have been established to improve the skills of medical technologists. Knowledge about clinical chemistry and the skills necessary for quality control should be obtained through these qualifications. Quality control is required to keep medical treatment fees low and also contributes to hospital management.
In Japan, the management of toxic, highly poisonous, and hazardous substances is strictly regulated. These substances should be similarly handled in hospitals and clinical laboratories. Thus, paramedics should have chemical knowledge about these substances to allow them to handle them appropriately.
We thank my colleagues at Kyoto University, Osaka University, Kyushu University, and Kyoto Institute of Technology for their help with this study. This study was partially supported by the Japan Leukemia Research Fund (H.K).